Redox‐Active NOx Ligands in Palladium‐Mediated Processes - Fairlamb - 2015 - Angewandte Chemie International Edition - Wiley Online Library

Exploiting Noninnocent (E,E)‐Dibenzylideneacetone (dba) Effects in Palladium(0)‐Mediated Cross‐Coupling Reactions: Modulation of the Electronic Properties of dba Affects Catalyst Activity and Stability in Ligand and Ligand‐Free Reaction Systems ...

Halide and pseudohalide effects in Pd-catalysed cross-coupling reactions - New Journal of Chemistry (RSC Publishing) DOI:10.1039/B605699G

Exploiting Noninnocent (E,E)‐Dibenzylideneacetone (dba) Effects in Palladium(0)‐Mediated Cross‐Coupling Reactions: Modulation of the Electronic Properties of dba Affects Catalyst Activity and Stability in Ligand and Ligand‐Free Reaction Systems ...

Exploiting Noninnocent (E,E)‐Dibenzylideneacetone (dba) Effects in Palladium(0)‐Mediated Cross‐Coupling Reactions: Modulation of the Electronic Properties of dba Affects Catalyst Activity and Stability in Ligand and Ligand‐Free Reaction Systems ...

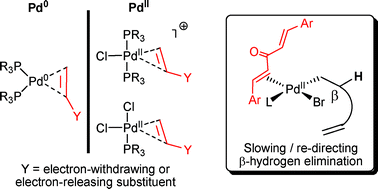
Palladium‐Catalysed Cross‐Coupling and Related Processes: Some Interesting Observations That Have Been Exploited in Synthetic Chemistry - McGlacken - 2009 - European Journal of Organic Chemistry - Wiley Online Library
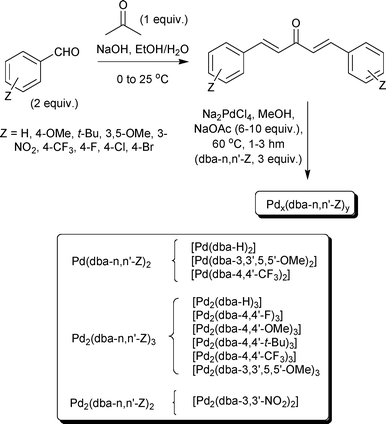
π-Acidic alkene ligand effects in Pd-catalysed cross-coupling processes: exploiting the interaction of dibenzylidene acetone (dba) and related ligand ... - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B811772A

Analysis of Stereochemical Convergence in Asymmetric Pd‐Catalysed Allylic Alkylation Reactions Complicated by Halide and Memory Effects - Fairlamb - 2002 - Chemistry – A European Journal - Wiley Online Library
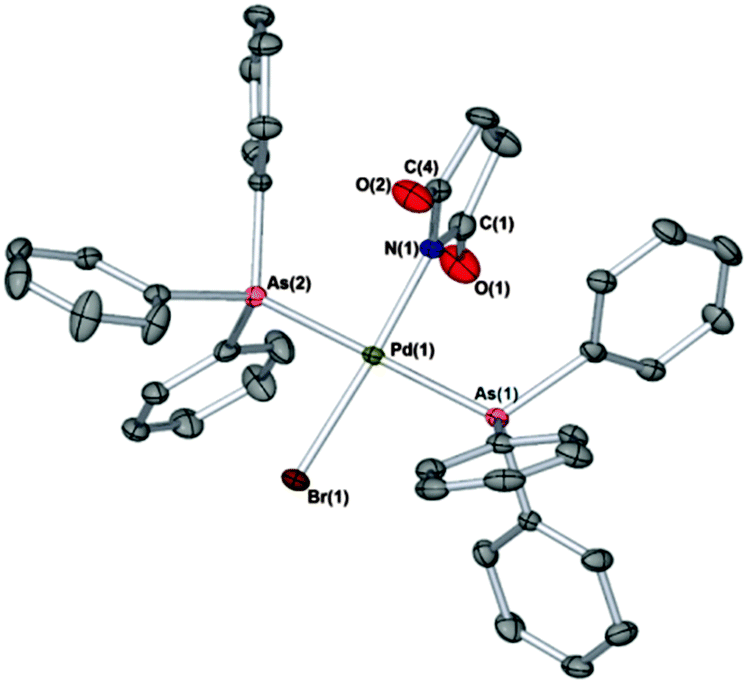
AsCat and FurCat: new Pd catalysts for selective room-temperature Stille cross-couplings of benzyl chlorides with organostannanes - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC09810B

Palladium‐Catalysed Cross‐Coupling and Related Processes: Some Interesting Observations That Have Been Exploited in Synthetic Chemistry - McGlacken - 2009 - European Journal of Organic Chemistry - Wiley Online Library

Figure 15 from The elusive structure of Pd2(dba)3. Examination by isotopic labeling, NMR spectroscopy, and X-ray diffraction analysis: synthesis and characterization of Pd2(dba-Z)3 complexes. | Semantic Scholar