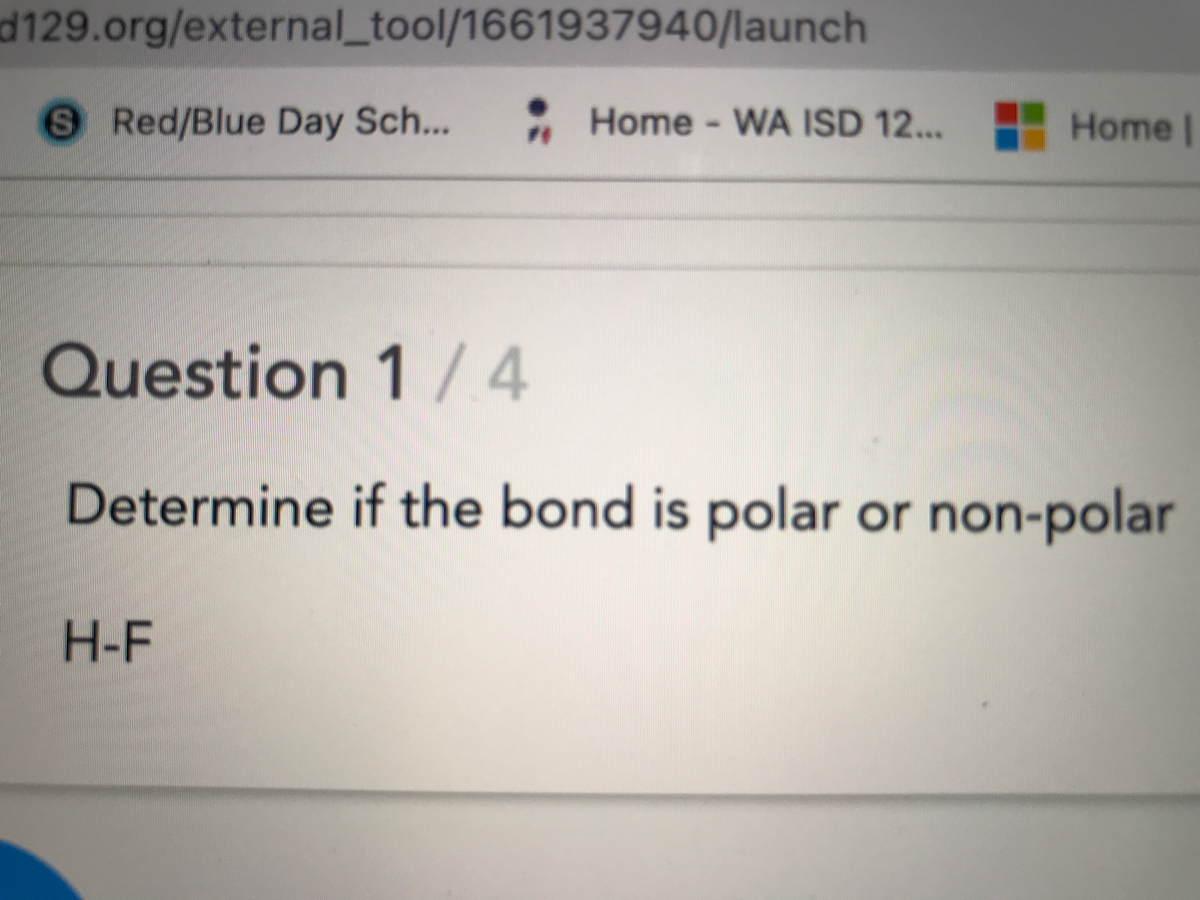
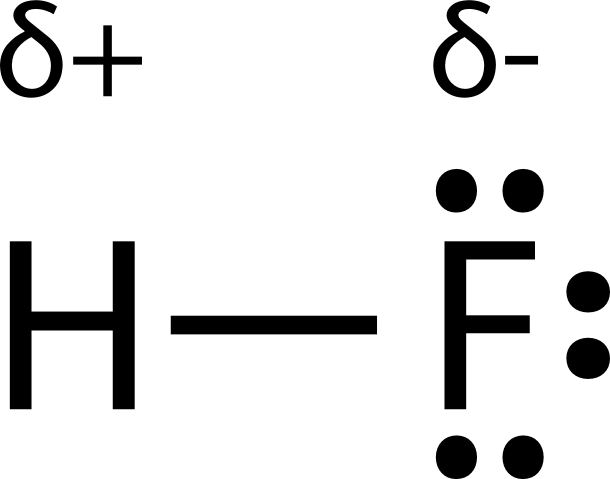Why is it so that the dipole moment of HI < HBr< HCl<HF, where HI is lowest and HF is highest? - Quora

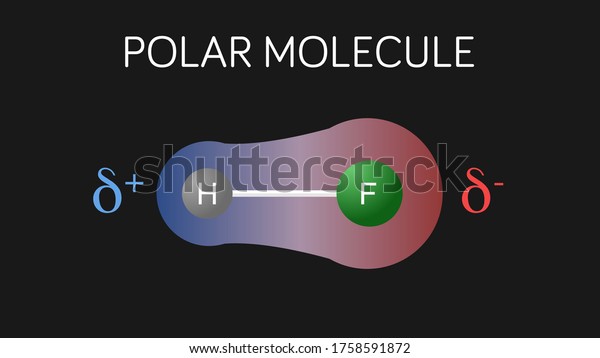
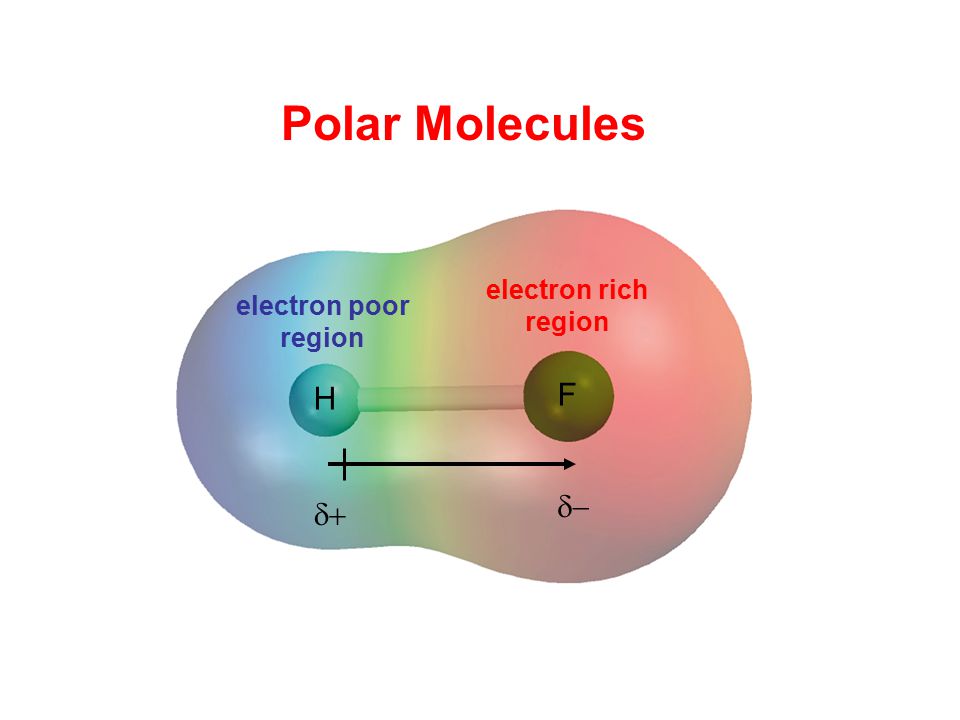
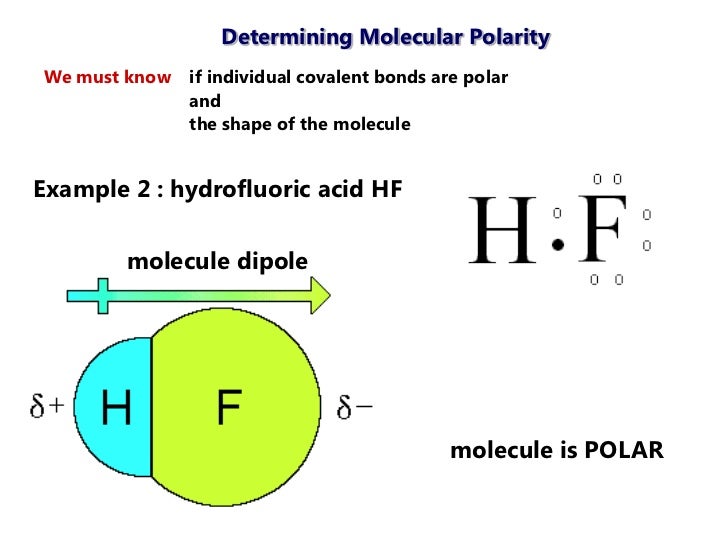
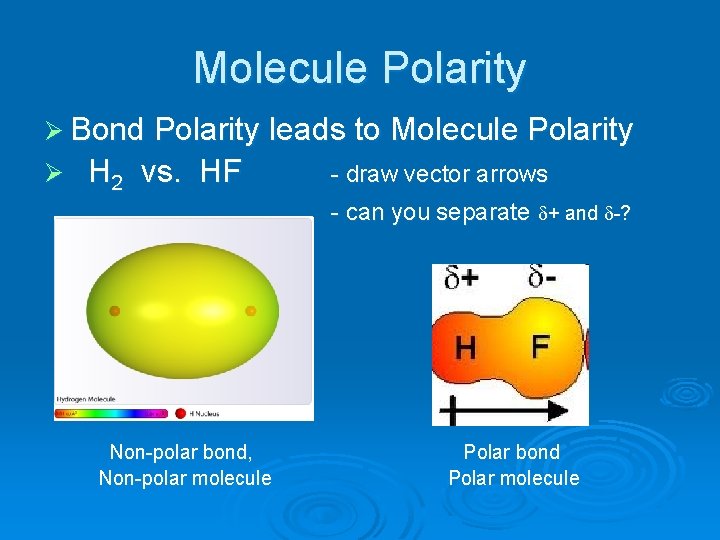
Chapter 8: Covalent Compounds Polarity A molecule, such as HF, that has a center of positive charge and a center of negative charge is said to be polar, - ppt download
HF equivalent resistance R d h in polar coordinate with an electrical... | Download Scientific Diagram

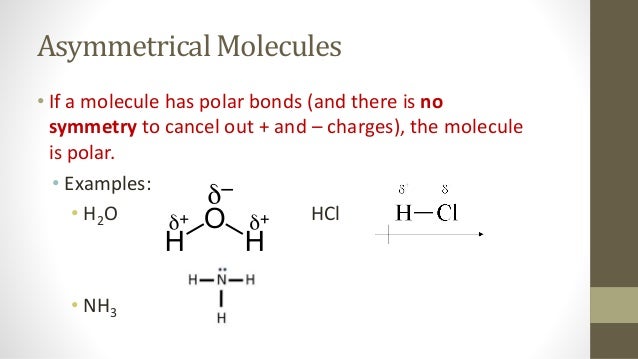
1 Molecular Polarity. 2 Polar Molecules Polar molecules are molecules that have a slightly positive end or pole and a slightly negative end or pole. The. - ppt download